Clinical trials are a cornerstone in the development of new interventions and therapies from pharmaceuticals to digital health applications. Clinical trials provide scientific evidence on safety and efficacy, and they inform clinical care. Despite being a staple of the R&D process for decades, traditional clinical trials are slow, expensive, and inefficient.
An emerging trend in the clinical trial landscape is the incorporation of digital health technologies into study design, such as mobile apps and remote monitoring devices. Digital health technology offers a way for clinical trials to be conducted virtually, referred to as decentralized, virtual, or siteless clinical trials. Once seen as a gimmick, virtual clinical trials are now commonplace and increasing in popularity.
Virtual clinical trials, and hybrid models, have the potential to cut costs, improve participant recruitment and retention, and can enable participants to join studies that otherwise would not, such as those with mobility issues or those who live in rural areas. Additionally, digital health technologies offer real-time and continuous data collection which provides a deeper understanding of a person’s condition against periodic data collection during site visits.

Pattern Health’s Digital Health Platform
Pattern Health helps researchers navigate the quickly evolving virtual clinical trials ecosystem. Our scalable and secure, no-code digital health platform enables researchers to conduct fully virtual and hybrid clinical trials, ultimately improving upon the cumbersome and inefficient models of the past. Our solution offers the adaptability and scale to support virtually any clinical trial that could benefit from a virtual or hybrid trial design. Pattern Health is HITRUST certified and compliant with HIPAA, GDPR, and FISMA regulations to ensure the privacy and security of research studies. Additionally, Pattern Health is 21 CFR Part 11 compliant, meeting the FDA’s requirements for traceability, validation, assurance, and auditability.
We partner with researchers to create digital health applications that power virtual trials from patient screening to remote data collection and the delivery of behavioral interventions. A growing network of leading academic medical centers, including 12 of the top 20, are using our platform to power studies and digital health innovation initiatives. Here’s why we are their platform of choice for virtual clinical trials:
Patient screening, enrollment, consent, and onboarding.
Pattern Health offers flexible user interfaces and workflows for the onboarding of new patients, including mobile and web-based experiences. We can support the process from beginning to end, or connect into existing workflows as needed. Our enrollment feature supports the randomized assignment of patients to study groups. We offer integrations with Hellosign for 21 CFR Part 11-compliant e-consent and StudyPages for recruiting and enrollment.
Remote data collection.
Pattern Health supports manual and passive data collection. Our platform can collect passive data from integrated, third-party devices and apps, such as Fitbit (exercise and sleep tracking), Nutrionix (food logging), Apple HealthKit (biometrics), and more. We currently offer 20+ integrations that are ready for use “off the shelf”, with more coming soon. Our development team can also build custom integrations by request. Additionally, we can collect data directly through our mobile and web apps, such as the active entry of biometrics, med adherence, survey responses, patient-reported outcomes, and more.
Delivery of digital health interventions.
Pattern Health’s digital health platform specializes in the delivery and monitoring of behavioral interventions. We partner with researchers to develop custom web and mobile apps for patients that deliver reminders, education, and communication tools. Our platform is highly flexible and can be used for a wide range of conditions and use cases.
Incentives and payments.
Pattern Health offers a full-featured solution to help study teams implement their incentive strategy. Incentives include automated payments, motivational messages, and manually delivered incentives such as gift cards. Pattern Health supports Paypal and Venmo for payment processing of automated incentives and offers a manual point tracking system for those who want to track incentive eligibility in the platform but issue rewards using their own process.
Reporting and data sharing.
Pattern Health can collect and aggregate data from many sources and offers a number of ways to access and report on results. Our web console provides access to self-service reporting tools and allows the manual export of data in common research formats. Additionally, we offer an API to pull data into whatever analysis software you need, such as R or SAS.
UC Davis’ GROWell Use Case
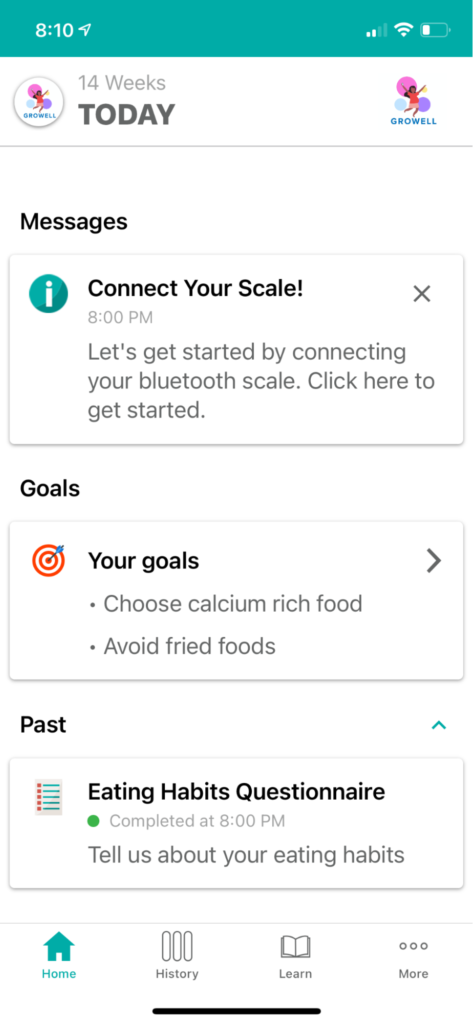
GROWell is a study designed to learn whether a mobile health program powered by Pattern Health’s platform can help people to have healthy pregnancies and return to pre-pregnancy health after giving birth.
The GROWell study uses Pattern Health to power a completely virtual clinical trial. The study leverages our integration with StudyPages to advertise, recruit, screen, and collect consent from study participants. Once screened, participants are automatically enrolled and configured as study participants on the Pattern Health mobile app. After the enrollment process, participants become a part of the approximately 12-month behavioral intervention program, which is delivered in the form of educational materials and surveys through the Pattern Health app as well as text messages.